

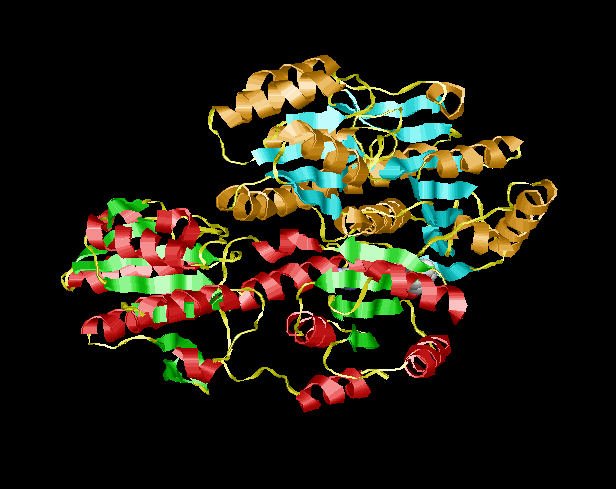
There are two binding sites on the enzymes Phosphofructokinase. The Active site where the groups transfer chemistry occurs and the "allosteric site". The latter only functions in binding molecules and performs NO chemistry. It does, however, bind a variety of molecules each of which effects the equilibrium of Good (R) to Poor (T) structural forms of the protein. The concetration of the "good form" of the protein is increased when ADP or fructose 2,6 bisphosphate is bound to the allosteric site and ALSO by fructose-6-phosphate binding at the ACTIVE SITE. The concentration of the Poor structural form of the protein is increased when ATP, phosphoglycolic acid (PGA) or citrate bind to the allosteric site.
|
|
|
Ribbon Structure only of the Good (R) form of Phosphofructokinase | |