

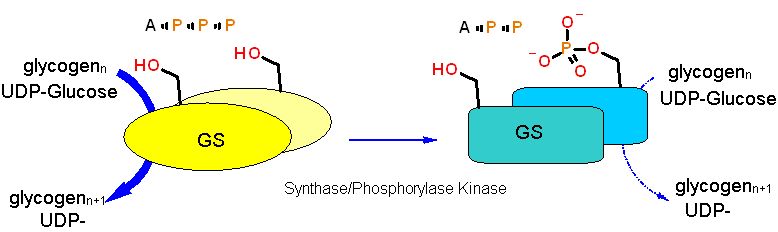
 When a protein is covalently modified it is generally at a specific place and on a specific amino acid sidechain and of course requires an enzyme to catalyze the modification. Here the target protein, Glycogen Synthease (in this case) is phosphorylated on a spcific serine by the enzyme "Synthase/Phosphorylase kinase" using ATP. Glycogen Synthase shifts from a structure that has GOOD catalytic properties (Thick blue arrow) to one that has POOR catalytic properties (thin blue dashed arrow) when it is modified. There are several serines that can be modified in glycogen synthase. Other proteins can be phosphorylated on serine as well as threonine or even tyrosine.
When a protein is covalently modified it is generally at a specific place and on a specific amino acid sidechain and of course requires an enzyme to catalyze the modification. Here the target protein, Glycogen Synthease (in this case) is phosphorylated on a spcific serine by the enzyme "Synthase/Phosphorylase kinase" using ATP. Glycogen Synthase shifts from a structure that has GOOD catalytic properties (Thick blue arrow) to one that has POOR catalytic properties (thin blue dashed arrow) when it is modified. There are several serines that can be modified in glycogen synthase. Other proteins can be phosphorylated on serine as well as threonine or even tyrosine.
Covalent Modification (Phosphorylation in this case) - just like a compound binding at the allosteric site - stabilizes a specific structural form of an enzyme. And also just like a compound binding to an allosteric site, alters the equilibrium between GOOD and POOR forms altering the enzyme efficiency accordingly.
| Modification | Enzyme | Stabilizes | Effect |
| Phosphorylation | Glycogen synthase | POOR (T) | Decreases glycogen synthesis AND increases glycogen breakdown |
| Glycogen Phosphorylase | GOOD (R) | ||
| Phosphorylation | Phosphofructokinase-2 | POOR (T) | decreases glycolysis and increases gluconeogenesis |
| Fructose 2,6 Bisphosphatase | GOOD (R) | ||
| These latter two activities are on the same polypeptide this configuration leads to hydrolysis of fructose-2,6-Bisphosphate to fructose-6-phosphate. Effectively increases the gluconeogenesis rates | |||
The same information put into graphic form.....
| HIGH Blood Sugar | LOW Blood Sugar |
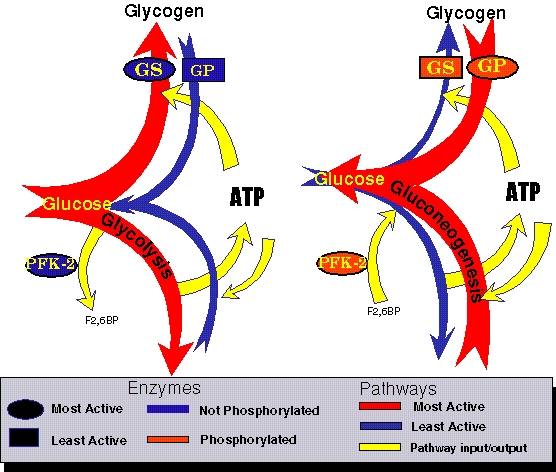 | |
| This scenario is specific to Liver Cells. On the left is the case for high blood sugar where glucose is stored as glycoge. This requires energy input for each and every glucose added. So it is essential that glycolysis also function to generate the necessary ATP. This is represetnted with the large red arrows going from glucose to glycogen and glycolysis. In the opposite case, when blood sugar is low, Glucagon is secereted and a variety of proteins are phosphorylated. The result is the large red arrows showing that glycogen is degraded and gluconeogenesis is the most effective pathway. This ensures the most effective way to ensure that glucose is generated and secreted out to the blood. | |
Liver cells respond to organismal requirements as well as the internal metabolic state. Many cell types respond to insulin (released by the pancreas in response to increasing blood glucose levels), which induces glucose uptake from the blood. Liver cells are also induced to increase Glycogen synthesis and glycolysis by insulin. Liver cells also respond to Glucagon (released by the pancreas in response to low blood glucose levels) by increasing both Glycogen breakdown and gluconeogenesis so that glucose can be shipped out to the circulation system. The regulation of the enzymes is accomplished by a series of covalent modification events that leads to the phosphorylation of GS (to inactivate) GP (to activate) and PFK-2 (activates the F26BPase activity and inhibits the PFK-2 activity)
Keep in mind that the allosteric regulation events are ALSO still effective for all of the enzymes shown.